 
McCulloh gives a few of what he calls the "more interesting examples" of the application of these laws in extent and utility. Nor should it, therefore, be a matter of surprise that already, in the short space of time, not yet one generation, elapsed since the mechanical theory of heat has been freely adopted, whole branches of physical science have been revolutionized by it. When we reflect how generally physical phenomena are connected with thermal changes and relations, it at once becomes obvious that there are few, if any, branches of natural science which are not more or less dependent upon the great truths under consideration. what are currently known as the first law of thermodynamics and the second law of thermodynamics, are innumerable: McCulloh then declares that the applications of these two laws, i.e. In 1876, American civil engineer Richard Sears McCulloh, in his Treatise on the Mechanical Theory of Heat and its Application to the Steam-Engine, which was an early thermodynamics textbook, states, after speaking about the laws of the physical world, that "there are none that are established on a firmer basis than the two general propositions of Joule and Carnot which constitute the fundamental laws of our subject." McCulloh then goes on to show that these two laws may be combined in a single expression as follows: The general struggle for existence of animate beings is not a struggle for raw materials – these, for organisms, are air, water and soil, all abundantly available – nor for energy which exists in plenty in any body in the form of heat, but a struggle for entropy, which becomes available through the transition of energy from the hot sun to the cold earth. In 1875, building on the works of Clausius and Kelvin, Boltzmann reasoned: Building on this, one of the first to speculate on a possible thermodynamic perspective of organic evolution was the Austrian physicist Ludwig Boltzmann. In 1863, Rudolf Clausius published his noted memoir On the Concentration of Rays of Heat and Light, and on the Limits of Its Action, wherein he outlined a preliminary relationship, based on his own work and that of William Thomson (Lord Kelvin), between living processes and his newly developed concept of entropy. Ideas about the relationship between entropy and living organisms have inspired hypotheses and speculations in many contexts, including psychology, information theory, the origin of life, and the possibility of extraterrestrial life. 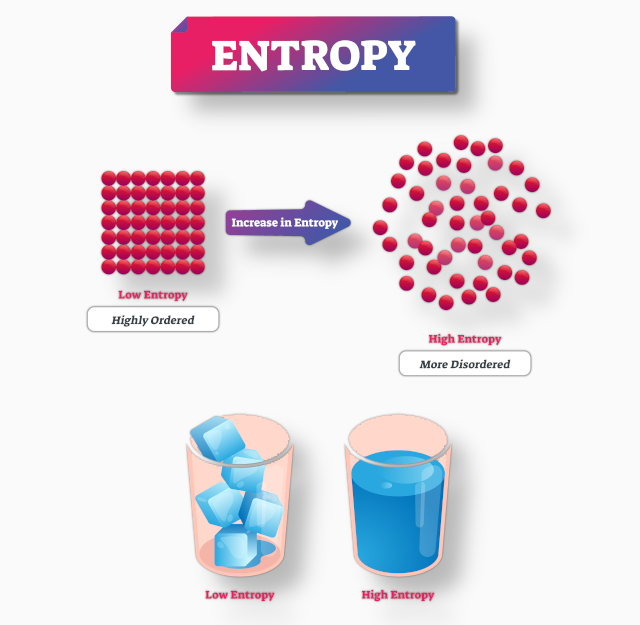
More recent work has restricted the discussion to Gibbs free energy because biological processes on Earth normally occur at a constant temperature and pressure, such as in the atmosphere or at the bottom of the ocean, but not across both over short periods of time for individual organisms. In his book, Schrödinger originally stated that life feeds on negative entropy, or negentropy as it is sometimes called, but in a later edition corrected himself in response to complaints and stated that the true source is free energy. The 1944 book What is Life? by Nobel-laureate physicist Erwin Schrödinger stimulated further research in the field. In 1910, American historian Henry Adams printed and distributed to university libraries and history professors the small volume A Letter to American Teachers of History proposing a theory of history based on the second law of thermodynamics and on the principle of entropy. Research concerning the relationship between the thermodynamic quantity entropy and both the origin and evolution of life began around the turn of the 20th century. 
Relationship between the thermodynamic concept of entropy and the evolution of living organisms 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
